The European Medicines Agency (EMA) has initiated a series of actions to respond to the ongoing monkeypox outbreak, which has been escalated by the World Health Organization (WHO) to a Public Health Emergency of International Concern (PHEIC) on Saturday 23 July. This is the first new PHEIC to be declared since the regulation reinforcing EMA’s role in crisis preparedness and management of medicinal products and medical devices has become applicable.
Since the start of the recent monkeypox outbreak, EMA has been monitoring the situation closely and already taken multiple actions to prepare for and support the EU response. This includes the Agency’s recommendation on 22 July to approve an extension of indication for the vaccine Imvanex to protect adults from monkeypox disease. The new powers given to the Agency under its extended mandate trigger additional activities now that monkeypox has been declared a public health emergency.
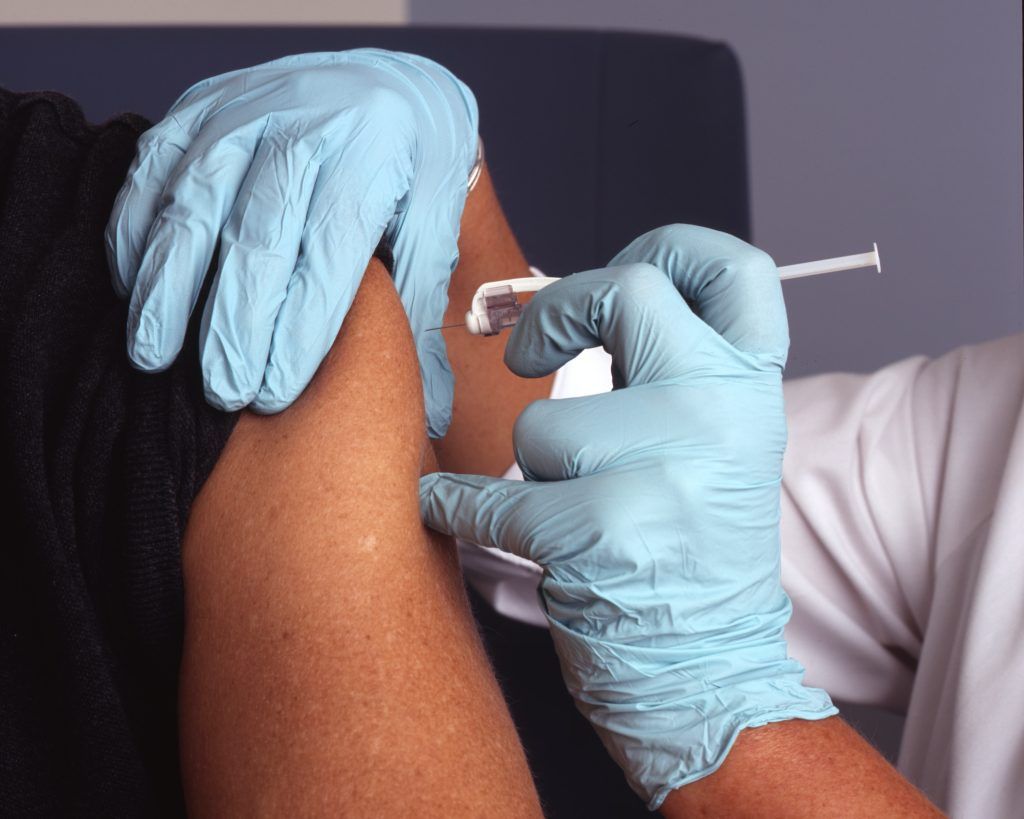
Start of delivery of vaccines in response to the monkeypox outbreak
|
Monitoring of supply, demand and shortages of medicinal products
The EMA Executive Steering Group on Shortages and Safety of Medicinal Products (MSSG), established by the new Regulation, will produce and maintain a formal list of critical medicines for the monkeypox public health emergency. The list will be drawn up in a collaborative process involving Member States, healthcare professionals, patients and consumers.
Marketing authorisation holders of medicines included in the list will be required to regularly update EMA with relevant information on potential or actual shortages and available stocks, forecasts of supply and demand. In addition, Member States will provide regular reports on estimated demand for these medicines at national level.
This will enable the MSSG to recommend and coordinate appropriate EU-level actions to the European Commission and EU Member States in order to prevent or mitigate potential or actual shortages of critical medicines to safeguard public health.
Evolution of monkeypox in Europe
|
There are currently two medicinal products authorised specifically for monkeypox in the EU:
- The medicine Tecovirimat SIGA is authorised for use to treat smallpox, monkeypox and cowpox.
- The vaccine Imvanex is authorised to protect adults against smallpox and monkeypox. Imvanex is marketed as Jynneos in the US and considering the limited availability of Imvanex, the ETF has recommended that Jynneos can be used to provide protection against monkeypox disease in the EU.
More information: European Medicine Agency – Press release






Leave a Reply