Today the European Commission is proposing to create a Digital Green Certificate to facilitate safe free movement inside the EU during the COVID-19 pandemic. The Digital Green Certificate will be a proof that a person has been vaccinated against COVID-19, received a negative test result or recovered from COVID-19. It will be available, free of charge, in […]
European Commission authorises fourth vaccine against COVID-19: Janssen
The European Commission has granted a conditional marketing authorisation (CMA) for the COVID‑19 vaccine developed by Janssen Pharmaceutica NV, one of the Janssen Pharmaceutical Companies of Johnson & Johnson, and the fourth COVID-19 vaccine authorised in the EU. This authorisation follows a positive scientific recommendation based on a thorough assessment of the safety, effectiveness and […]
Will the Spanish economy be boosted after Covid-19?
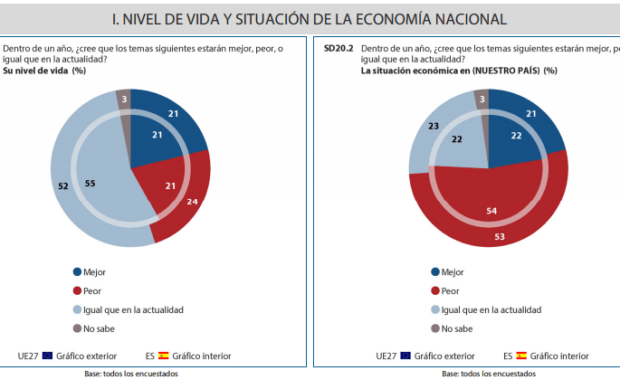
The pandemic has affected not only people’s health, but the economy has also suffered a huge decline in most countries. It is believed that the European Union is in charge of carrying out this mission within Europe, citizens agree that practical solutions must be put forward to combat this crisis. In a survey conducted by […]
EMA receives application for conditional marketing authorisation of COVID-19 Vaccine Janssen
EMA has received an application for conditional marketing authorisation (CMA) for a COVID-19 vaccine developed by Janssen-Cilag International N.V. EMA’s human medicines committee (CHMP) will assess the vaccine, known as COVID-19 Vaccine Janssen, under an accelerated timetable. The Committee could issue an opinion by the middle of March 2021, provided the company’s data on the vaccine’s efficacy, safety […]
EMA preparing guidance to tackle COVID-19 variants
EMA is developing guidance for manufacturers planning changes to the existing COVID-19 vaccines to tackle the new virus variants. In order to consider options for additional testing and development of vaccines that are effective against new virus mutations, the Agency has requested all vaccine developers to investigate if their vaccine can offer protection against any […]
COVID-19: Council updates recommendation on measures affecting free movement
The Council adopted a recommendation amending the recommendation on a coordinated approach to the restrictions of free movement in response to the COVID-19 pandemic. Under the updated recommendation, a new colour (dark red) is added to the existing categories of green, orange, red and grey in the weekly map published by the ECDC. This colour […]
COVID-19 Evolution – 2020
This document collects and analyzes the data on the evolution of COVID-19, collected by the Johns Hopkins University and published daily by the European Documentation Center of Almeria during the year 2020. On January 24, 2020 France reported the first case of COVID-19, being the first case, being the first known case of COVID-19 in […]
Plastics, a growing environmental and climate concern: how can Europe revert that trend?
The ever-increasing amount of plastic, its impact on biodiversity and contribution to climate change, and how to deal with it in a circular economy perspective have been on the European Union’s policy agenda for years. The COVID-19 pandemic has only increased the attention for plastic waste with images of masks in our seas, and large […]
MEPs urge EU countries to be transparent about their COVID-19 vaccine supplies
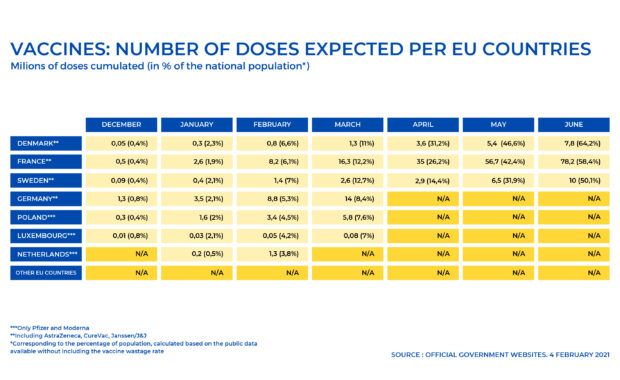
Parliament’s Environment, Public Health and Food Safety Committee calls for a fact-based approach to the EU vaccines rollout to avoid disinformation. In order to respond to European citizens’ growing concerns, data on the number of vaccine doses supplied and on the vaccination schedules for each country must be transparent and provided on a monthly basis […]