EMA is aware that the Danish Health Authority has paused its vaccination campaign with COVID-19 Vaccine AstraZeneca. This was decided as a precautionary measure while a full investigation is ongoing into reports of blood clots in people who received the vaccine, including one case in Denmark where a person died. Some other Member States have […]
European Commission authorises fourth vaccine against COVID-19: Janssen
The European Commission has granted a conditional marketing authorisation (CMA) for the COVID‑19 vaccine developed by Janssen Pharmaceutica NV, one of the Janssen Pharmaceutical Companies of Johnson & Johnson, and the fourth COVID-19 vaccine authorised in the EU. This authorisation follows a positive scientific recommendation based on a thorough assessment of the safety, effectiveness and […]
EMA receives application for conditional marketing authorisation of COVID-19 Vaccine Janssen
EMA has received an application for conditional marketing authorisation (CMA) for a COVID-19 vaccine developed by Janssen-Cilag International N.V. EMA’s human medicines committee (CHMP) will assess the vaccine, known as COVID-19 Vaccine Janssen, under an accelerated timetable. The Committee could issue an opinion by the middle of March 2021, provided the company’s data on the vaccine’s efficacy, safety […]
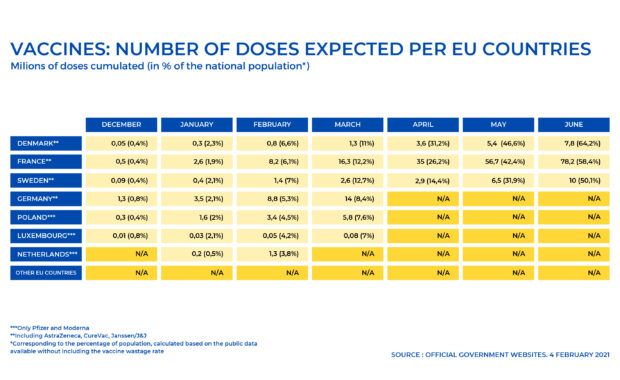
MEPs urge EU countries to be transparent about their COVID-19 vaccine supplies
Parliament’s Environment, Public Health and Food Safety Committee calls for a fact-based approach to the EU vaccines rollout to avoid disinformation. In order to respond to European citizens’ growing concerns, data on the number of vaccine doses supplied and on the vaccination schedules for each country must be transparent and provided on a monthly basis […]
European Commission authorises the AstraZeneca vaccine against COVID-19
The European Commission has granted a conditional marketing authorisation (CMA) for the COVID‑19 vaccine developed by AstraZeneca, the third COVID-19 vaccine authorised in the EU. This authorisation follows a positive scientific recommendation based on a thorough assessment of the safety, effectiveness and quality of the vaccine by the European Medicines Agency (EMA) and is endorsed […]
EU urges AstraZeneca to explain vaccine delivery delays
▶️ Watch the press conference by Commissioner @SKyriakidesEU on vaccine deliveries and on the export transparency scheme. #StrongerTogether https://t.co/iczvKiAiRm — European Commission 🇪🇺 (@EU_Commission) January 25, 2021 Last Friday, the company AstraZeneca surprisingly informed the Commission and the European Union Member States that it intends to supply considerably fewer doses in the coming weeks than […]
EMA receives application for conditional marketing authorisation of COVID-19 Vaccine AstraZeneca
EMA has received an application for conditional marketing authorisation (CMA) for a COVID-19 vaccine developed by AstraZeneca and Oxford University. The assessment of the vaccine, known as COVID-19 Vaccine AstraZeneca, will proceed under an accelerated timeline. An opinion on the marketing authorisation could be issued by 29 January during the meeting of EMA’s scientific committee for human […]
European Commission authorises second safe and effective vaccine against COVID-19
The European Commission has granted a conditional marketing authorisation (CMA) for the COVID‑19 vaccine developed by Moderna, the second COVID-19 vaccine authorised in the EU. This authorisation follows a positive scientific recommendation based on a thorough assessment of the safety, effectiveness and quality of the vaccine by the European Medicines Agency (EMA) and is endorsed […]
European Commission authorises first safe and effective vaccine against COVID-19
The European Commission has granted a conditional marketing authorisation (CMA) for the COVID‑19 vaccine developed by BioNTech and Pfizer, making it the first COVID-19 vaccine authorised in the EU. This authorisation follows a positive scientific recommendation based on a thorough assessment of the safety, effectiveness and quality of the vaccine by the European Medicines Agency […]