The EU and AstraZeneca have reached an agreement which will secure the delivery of the remaining COVID-19 vaccine doses to Member States under the terms of the Advance Purchase Agreement concluded on 27 August 2020 with AstraZeneca. The agreement will also end the pending litigation before the Brussels Court. Commissioner for Health and Food Safety […]
AstraZeneca’s vaccine: benefits and risks
Vaxzevria (formerly COVID-19 Vaccine AstraZeneca) is authorised in the EU to prevent COVID-19, which can cause severe disease and death. The disease can also have long-term consequences in people of all ages, including in otherwise healthy people. The benefits of Vaxzevria outweigh its risks in adults of all age groups; however, very rare cases of […]
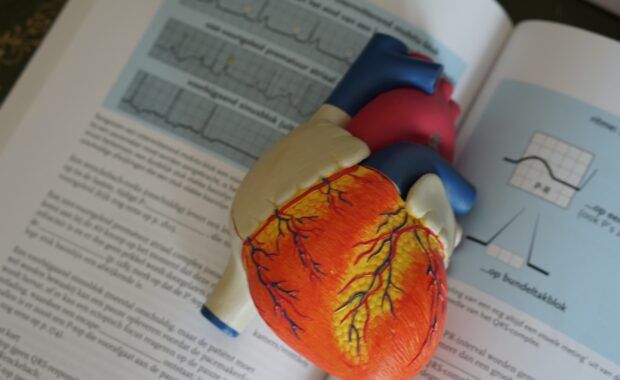
EMA finds possible link to very rare cases of unusual blood clots with low blood platelets
EMA confirms overall benefit-risk remains positive EMA’s safety committee (PRAC) has concluded today that unusual blood clots with low blood platelets should be listed as very rare side effects of Vaxzevria (formerly COVID-19 Vaccine AstraZeneca). In reaching its conclusion, the committee took into consideration all currently available evidence, including the advice from an ad hoc […]
COVID-19 Vaccine AstraZeneca: PRAC investigating cases of thromboembolic events
EMA is aware that the Danish Health Authority has paused its vaccination campaign with COVID-19 Vaccine AstraZeneca. This was decided as a precautionary measure while a full investigation is ongoing into reports of blood clots in people who received the vaccine, including one case in Denmark where a person died. Some other Member States have […]
European Commission authorises the AstraZeneca vaccine against COVID-19
The European Commission has granted a conditional marketing authorisation (CMA) for the COVID‑19 vaccine developed by AstraZeneca, the third COVID-19 vaccine authorised in the EU. This authorisation follows a positive scientific recommendation based on a thorough assessment of the safety, effectiveness and quality of the vaccine by the European Medicines Agency (EMA) and is endorsed […]
EU urges AstraZeneca to explain vaccine delivery delays
▶️ Watch the press conference by Commissioner @SKyriakidesEU on vaccine deliveries and on the export transparency scheme. #StrongerTogether https://t.co/iczvKiAiRm — European Commission 🇪🇺 (@EU_Commission) January 25, 2021 Last Friday, the company AstraZeneca surprisingly informed the Commission and the European Union Member States that it intends to supply considerably fewer doses in the coming weeks than […]